A battery is a device that stores chemical energy and converts it into electrical energy to power electronic devices. At its core, a battery consists of one or more electrochemical cells connected in series or parallel. Each cell features an anode, cathode및 electrolyte, which work together to create a potential difference and generate electric current when connected to a load.
When a battery supplies power, ions travel through the electrolyte while electrons flow through the external circuit — this flow of electrons is what we recognize as usable electrical energy. Batteries are ubiquitous in today’s world, from small AA cells to large lead-acid car batteries and advanced lithium-ion packs for electric vehicles.
Understanding basic battery principles — including 전압, capacity, 에너지 밀도및 chemical reactions — helps consumers make informed choices and extends device longevity.
목차
Why Are There Different Types of Batteries?
Battery technology isn’t one-size-fits-all. Different devices require different power characteristics, which has led to a variety of battery chemistries and formats.
Two of the most common primary (non-rechargeable) battery types are:
- Carbon-Zinc Batteries
- 알카라인 배터리
These batteries are widely used in household devices like flashlights, remote controls, clocks, and toys. Despite serving similar basic purposes, they differ significantly in performance, energy output, cost, and ideal applications.
What Is a Carbon-Zinc Battery?
탄소-아연 배터리, also known historically as Leclanché cells, were among the first dry cells widely used in the 20th century.
A typical carbon-zinc cell includes:
- A zinc can act as the anode
- A carbon rod as the cathode
- An acidic electrolyte such as ammonium chloride paste
The electrical reaction involves oxidation of zinc at the anode and reduction reactions at the cathode, generating a voltage of roughly 1.5 V per cell. These batteries are often labeled simply as “standard” or “everyday” batteries.
Advantages of Carbon-Zinc Batteries
- Economical and inexpensive to produce
- Good for low-drain applications
- Relatively lightweight and simple in design
Limitations of Carbon-Zinc Batteries
- Lower 에너지 밀도 compared to modern alternatives
- 수명 단축 and rapid voltage drop under load
- More prone to leakage, especially when depleted
- Performance drops in cold environments
Because of these drawbacks, carbon-zinc batteries have largely been replaced by alkaline cells in many applications.
What Makes Alkaline Batteries Better Than Carbon-Zinc?
알카라인 배터리 represent a significant advance in battery chemistry and design. Instead of acidic electrolytes, they use a 수산화 칼륨 electrolyte, which offers a more stable chemical environment.
Key components include:
- Zinc powder as the anode
- Manganese dioxide as the cathode
- Potassium hydroxide electrolyte
This improved chemistry results in higher energy density, more stable voltage output, better performance under moderate to heavy loads, and extended shelf life.
Major Benefits of Alkaline Batteries
- Longer operational life — often 2–3× that of carbon-zinc cells
- Better performance in medium to high-drain devices
- Enhanced leakage resistance due to improved sealing
- More consistent voltage delivery across the life of the battery
- Operate more reliably across a wide range of temperatures
These advantages make alkaline batteries the preferred choice for:
- Digital cameras
- Gaming controllers
- Portable radios
- High-performance flashlights
- Remote-controlled toys
Even though they cost more upfront than carbon-zinc cells, the longer life and reduced replacements often make alkaline batteries more cost-effective over time.
How Do Primary and Secondary Batteries Compare?
It’s important to distinguish between primary (non-rechargeable) 그리고 secondary (rechargeable) 배터리:
| 충전식 | ❌ No | ✅ Yes |
| 에너지 밀도 | Varies | High (e.g., Li-ion) |
| 비용 | Lower upfront | Higher upfront |
| 애플리케이션 | Low to medium drain | High demand + frequent use |
Alkaline and carbon-zinc batteries are both primary types — once discharged, they are typically discarded. In contrast, lead-acid, nickel-cadmium (NiCd), nickel-metal hydride (NiMH)및 lithium-ion batteries are examples of secondary types that recharge and reuse energy.
Where Do Lead-Acid and Lithium-Ion Fit In?
Although the earlier sections focus on primary dry cells, it’s worth briefly noting:
- Lead-acid batteries use lead and sulfuric acid to produce electricity and can be recharged, which is why they’re still widely used for automotive and backup power systems.
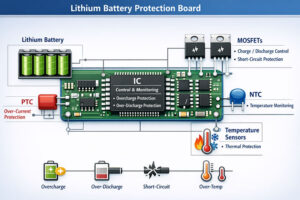
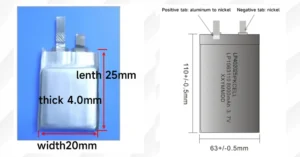
- 리튬 이온 배터리, common in smartphones and electric vehicles, offer extremely 높은 에너지 밀도 and excellent cycle life, though they require sophisticated battery management systems.
Why Does Battery Chemistry Matter for Consumers?
The choice of battery chemistry isn’t just academic — it affects real-world performance in several ways:
Runtime — High energy density means longer operation between replacements
비용 효율성 — Longer battery life reduces the total cost of ownership
Leak Resistance — Superior sealing protects devices
Temperature Performance — Some chemistries function better in extremes
For everyday use, alkaline cells often offer the best balance of performance and price. However, for professional tools or high-drain gadgets, advanced rechargeable batteries may be superior.
Conclusion: Choosing the Right Battery
Understanding differences in battery types — from classic carbon-zinc to modern alkaline and beyond — empowers smarter decisions and reduces wasteful replacements. Whether you’re powering a child’s toy or a high-drain device, knowing what battery chemistry matches your need ensures reliability, efficiency, and better overall performance.
Vedio from

 USB 충전식 리튬 배터리
USB 충전식 리튬 배터리